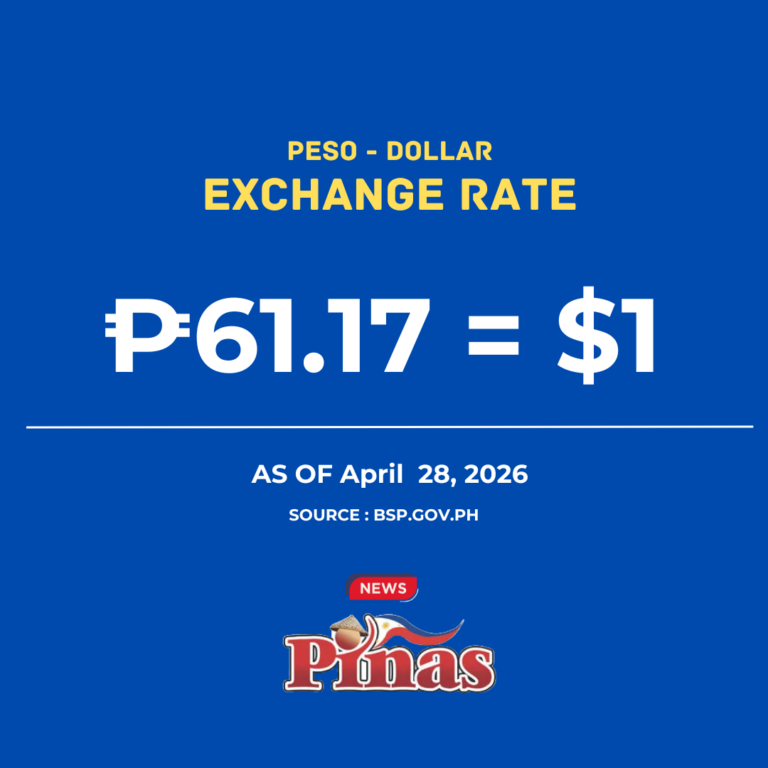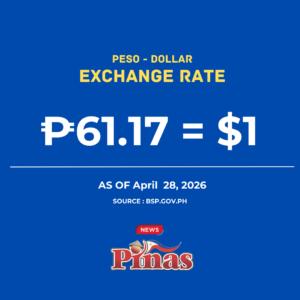The Quezon City Regional Trial Court (RTC) has denied government prosecutors’ bid to include transcripts of Senate inquiries into the anti-dengue Dengvaxia vaccine, as well as Food and Drug Administration (FDA) issuances, as evidence via judicial notice in connection with the reckless imprudence resulting to homicide case against former Health secretary Janette Garin and other ex-Health department officials due to utter lack of merit.
In a joint order dated November 14, the QC RTC said that while the inquiries were done by the Senate Blue Ribbon Committee, the Senate Committee on Health and the Senate Committee on Finance in aid of legislation from December 2017 to March 2018, the output of such committees cannot be taken in as truth at this point.
“To rule otherwise [in favor of the prosecution] would violate the right of all the accused to due process of law as they were not given the opportunity allotted to them by the Constitution to confront and assail the witness presented against them in the Committee hearings,” the joint order said.
The hearings’ findings “cannot be a substitute to the duty of the prosecution to prove the guilt of the accused beyond reasonable doubt based on competent and relevant evidence,” it added.
Garin’s co-accused in these cases include: Dr. Irma L. Asuncion, Dr. Julius A. Lecciones, Dr. Maria Rosario Z. Capeding, Melody Zamudio, Maria Lourdes Santiago, Dr. Mario Baquilod, Carlito Realuyo and Conchita Santos and Dr. Kenneth Hartigan-Go.
The court also noted that it already ruled that the transcripts of the Senate public hearings are inadmissible as evidence due to failure of the prosecution to attach them to the judicial affidavit of its witness, Dr. Fausto R. Quizon, Jr. and the incompetency of Dr. Anthony Leachon to testify on the same.
In addition, the court said it cannot take judicial notice of the Certificate of Product Registration BR – 1128 and BR – 1129 as well as FDA Circular No. 2013-004; FDA Decision dated December 29, 2017 and FDA Order dated December 21 2018 because these matters are “not of public knowledge and not capable of unquestionable demonstration.”
“Whether Sanofi Pasteur, Inc. (Dengavaxia manufacturer) failed to comply with the FDA requirements is a matter which must be proved through evidence during the trial of these cases. These documents cannot speak for itself,” the court said.
The court argued that testimonial evidence provides foundation for all types of evidence, while documentary evidence requires statement from the witness to make its way to the realm of admissible evidence.
“Simply put, to prove whether or not Sanofi Pasteur Inc. failed to comply with the FDA requirements, it is incumbent for the prosecution to prove that these documents are relevant and not excluded by rules of evidence testified to by a credible and competent witness,” the court said.
“Wherefore, premises considered, this Court denies the prosecution’s Motion to Take Judicial Notice of the Proceedings in Congress praying for this Court to take judicial notice of the: a) Blue Ribbon and Committee on Health and Demography and Finance inquiry, in aid of legislation, on 11 and 14 December 2017, 22 January 2018, 6 February 2018, and 13 March 2018; and the transcript of stenographic notes on said dates; b) Committee on Good Government and Public Accountability and the Committee on Health inquiry, in aid of legislation, on 26 February 2018, and the transcript of stenographic notes on said date; and c) Committee Report No. 368 and Committee Report No. 1336; and Motion to Take Judicial Notice of (a) Republic Act No. 3720, as amended; (b) Republic Act No. 9711; (c) Issuance of Certificate of Product Registration BR – 1128 and BR – 1129; (d) FDA Circular No. 2013-004; (e) FDA Decision dated 29 December 2017; and (0 FDA Order dated 21 December 2018″ for utter lack of merit,” the court added.
In November 2017, Dengvaxia manufacturer Sanofi-Pasteur announced that its vaccine posed a risk to those who were not infected by dengue prior to being injected with the vaccine.
This announcement, however, only came after the vaccine has already been administered to over 700,000 children and policemen.
Without admitting any wrongdoing, Sanofi returned P1.16 billion to the Department of Health for the unused doses of the vaccine. — BM, GMA Integrated News